Moles and Molarity
In chemistry, we often need a way to describe how much of a substance is present in a solution.
The mole is useful because it connects the tiny scale of atoms and molecules to measurable amounts in the laboratory,
while molarity tells us how concentrated a solution is by showing how many moles of solute are present in one litre of solution.
Molar units are widely used because they make it easier to calculate reacting amounts, prepare standard solutions, and carry out dilution calculations consistently.
Since volume can change with temperature, molarity is especially useful for everyday laboratory work, even though that also means it is temperature-dependent.
What is Molarity?
Key Equations
Number of Moles:
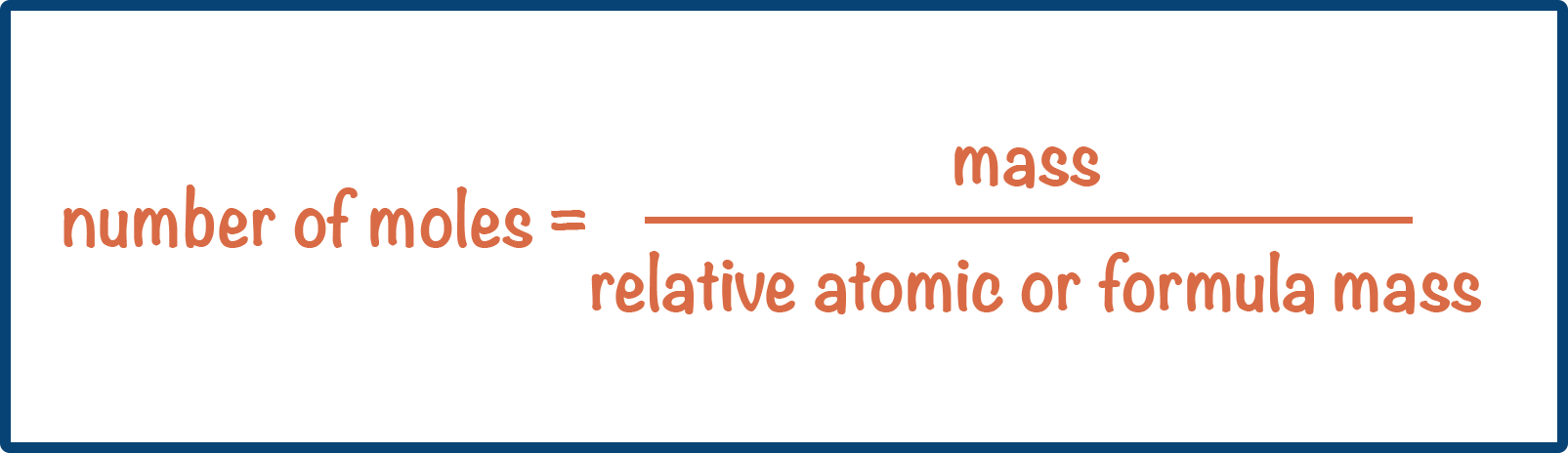
Number of Moles:

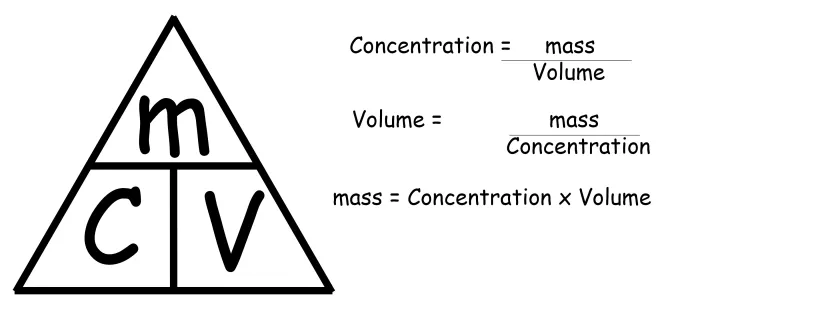
Molarity (Concentration in moles per L):
Video Guide
Paste your YouTube link here when ready:
Worked Examples
Example 1: Calculate molarity from moles and volume
A solution contains 0.50 mol of sodium chloride in 250 mL of solution.
Step 1: Convert volume to litres.
250mL = 0.250L (1000ml is equal to 1L)
Step 2: Use the molarity equation.
Molarity = no of moles / volume(L) = 0.5 / 0.25 = 2.0 mol/L
Answer: The molarity is 2.0 mol/L.
Example 2: Calculate moles from molarity and volume
A solution has a molarity of 0.20 mol/L and a volume of 150 mL.
Step 1: Convert volume to litres.
150mL = 0.150L (1L = 1000mL)
Step 2: Rearrange the equation.
no of moles = Concentration X Volume
n = 0.20 X 0.150 = 0.030 mol
Answer: The amount of solute is 0.030 mol.
Test Your Knowledge
Try each question before opening the answer.
1. What does the term molarity mean?
Answer: Molarity is the number of moles of solute per litre of solution.
2. Calculate the molarity of a solution containing 0.30 mol of solute in 600 mL of solution.
Answer: Convert 600 mL to 0.600 L, then use M = n / V.
M = 0.30 / 0.600 = 0.50 mol/L
3. How many moles are present in 2.5 L of a 0.40 mol/L solution?
Answer: Use n = M x V .
n = 0.40 x 2.5 = 1.0 mol
4. A student dissolves 5.85 g of sodium chloride in enough water to make 500 mL of solution. Calculate the molarity. [Mr of NaCl = 58.5]
Answer: First calculate moles:
n = 5.85 / 58.5 = 0.100 mol
Then convert 500 mL to 0.500 L and calculate molarity:
M = 0.100 / 0.500 = 0.200 mol/L
5. 25.0 mL of a 2.00 mol/L solution is diluted to 250 mL. What is the new concentration?
Answer: Use the dilution equation C_1 x V_1 = C_2 x V_2 .
(2.00)(25.0) = M_2(250)
C_2 = 0.200 mol/L
6. Explain why molarity is useful in titration calculations and solution preparation.
Answer: Molarity links the amount of solute to the volume of solution, making it ideal for measuring concentrations, preparing standard solutions, and calculating reacting amounts in titrations.
7. Challenge: A solution is made by dissolving 0.125 mol of glucose in 75.0 mL of solution. Calculate the molarity and then determine the volume needed to make 250 mL of a 0.200 mol/L solution from a stock of 0.500 mol/L.
Answer:
First part:
M = {0.125}{0.0750} = 1.67 mol/L
Second part:
C_1 x V_1 = C_2 x V_2
(0.500)V_1 = (0.200)(250)
V_1 = 100mL